What are the working principles and advantages of cyclone electrolysis technology?
1.1 Working principle
All electrolysis technologies are based on the basic theory of electrochemistry, and cyclone electrolysis technology is no exception. The traditional electrolysis technology is to place the cathode and anode in a slow flowing or stagnant tank. Under the effect of the electric field, the anions move directionally to the anode and the cations move directionally to the cathode. By controlling certain technical conditions, the metal cations to be obtained are electrodeposited at the cathode to obtain electrolytic products.
Cathodic reaction: metal ions get electrons at the cathode to form metal Me+(aq)+e - → Me (S)
Anodic reaction: The electrons obtained from the cathode need to be balanced by the loss of electrons from the anode. The anode has several possible reactions The main reaction is the oxidation of water in solution to generate oxygen, and the reaction is as follows: 2H2O → O2 (g)+4H++4e-
When the metal concentration in the electrolyte decreases, it is difficult to reduce the metal in the cathode without other reactions.
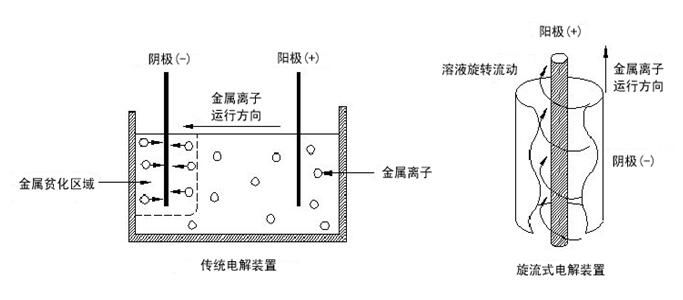
When the metal concentration is low The easy chemical reaction is the generation of hydrogen, as follows: 2H+(aq)+2e - → H2 (g)
Cyclone electrolysis technology is based on the difference of theoretical precipitation potential (E ¢) of each metal ion, that is, as long as the metal to be extracted has a large potential difference with other metal ions in the solution system, the metal with positive potential is easy to precipitate preferentially at the cathode. The key is to eliminate the adverse factors such as concentration polarization by telling the cyclone through the solution, It avoids the limitation of traditional electrolysis process affected by many factors (ion concentration, precipitation potential, concentration polarization, overpotential, pH value, etc.), and can produce high-quality metal products through simple technical conditions.
Taking the electrolysis (electrowinning) process of copper sulfate solution as an example, the reactions in the cyclone electrolysis device are as follows: direct current makes electrons transfer at the anode (positive pole) and cathode. The reaction on the anode consumes electrons (oxidation reaction), and the reaction on the cathode is the reaction to obtain electrons (reduction reaction).
2-valent copper ions are reduced to metal copper on the cathode surface: 2Cu2+(aq)+4e - | 2Cu (s) (1)
The electrons obtained at the cathode must be balanced with those lost at the anode. There are many possibilities of reactions occurring on the anode Possible oxygen evolution reaction of water. 2H2O(l)O2(g) + 4H+ + 4e- (2)
To sum up reaction 1 and reaction 2, the total reaction can be described as follows: Cu2+(aq)+H2O (l) I ½ O2 (g) + 2H+ + Cu(s) (3)

The current efficiency is mainly determined by other reactions besides the cathodic reduction of copper. Ideally, the reaction that takes place at the cathode is the reduction of copper ions to form copper metal at the cathode. If this is a cathodic reaction of..., the current is only used to deposit copper ions
When the concentration of copper in the electrolyte decreases, it is difficult to reduce copper at the cathode without other reactions. When the copper concentration is low The easy chemical reaction is the generation of hydrogen, as follows: 2H+(aq)+2e - → H2 (g)
1.2 Overview of cyclone electrolysis device
Cyclone electrolysis device is a comprehensive system integrating process equipment, electrical equipment and control system. The process is that the electrolytic solution is sent from the storage tank to the cyclone electrolytic tank arranged and arranged according to the design through the solution delivery pump. The solution circulates inside the device system. Valuable metal ions are separated from the electrolytic solution by selective electrolysis and adsorbed on the cathode plate. Barrel products are collected by special product collection device, and powder products are automatically collected by special powder collection system. Barrel products are further made into plate products through the subsequent supporting product processing devices. The matched intelligent power distribution device system and integrated control system, which can operate at high current density and... rate, adopt redundant hardware system and full digital software high-precision trigger regulation controller, duplex hot standby, anti harmonic interference, anti strong magnetic field interference, and communicate with the general control PLC system to jointly complete the intelligent control of electrolytic current. Combined with the patented technology of automatic control system, it can realize the independent detection of flow, liquid level, temperature, etc. in the production process, and ensure the accuracy and reliable operation in large-scale production.
1.3 Advantages of cyclone electrolysis technology
a. It is widely used in many industries;
b. Wide adaptability of raw materials, the same device can handle multiple metals, and selectively electrolyze and deposit metals;
c. Flexible and diversified, process design can be targeted according to customer needs;
d. Portable, modular components, easy to install, modular installation, small footprint, high space utilization;
e. Complete metal recovery and sharing, and ppm level metal recovery and separation;
f. It is easy to reduce the metal ions in the solution you need to recover to below 1000ppm, and make the valuable metals you need to recover into plate or powder products (>99.96%);
g. The solution is circulated in a closed circuit to effectively recover the acid in the solution and avoid the discharge of acid mist;
h. High current density and current efficiency;
i. Simplify the process, greatly reduce your operating costs and reduce your technical risks;
j. Compared with the traditional electrolysis technology, the cost recovery period can be at least 18 to 36 months ahead of time by using the cyclone electrolysis technology; Improve product purity, improve profitability, achieve rapid profitability, and have a higher rate of return on capital.
The article is reproduced on the Internet. If there is infringement, please contact to delete it.

Back to top